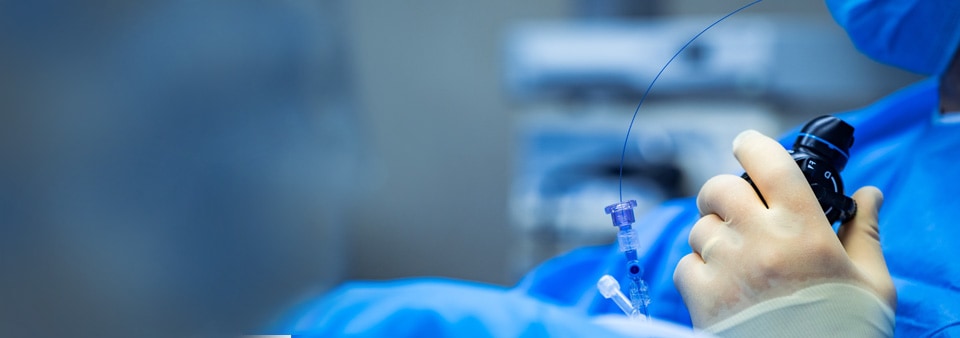
As a manufacturer of vascular medical devices, one of your biggest challenges in breaking into, or continuing your success in this market is navigating the regulatory process efficiently. Strong, statistically relevant clinical evidence demonstrating the safety and performance of your device to confirm clinical claims, intended use and intended patient population for the device is essential to ensure a successful outcome of your application.
Our vascular Technical Specialists are product experts who are highly trained in working with vascular medical device manufacturers and understand the specifics of these complex devices. We are experienced in working with apparatus, appliances, implants, instruments and other articles intended to be used in the vascular system. We also provide conformity assessments for medical devices for the heart itself used in the diagnosis, prevention, monitoring, treatment and alleviation of disease or injury as well as, in the investigation, replacement or modification of the anatomy in relation to the vasculature or the heart. Where additional expertise is required, we collaborate with our inhouse subject matter experts from all medical devices areas.