What is the CE marking transfer process?
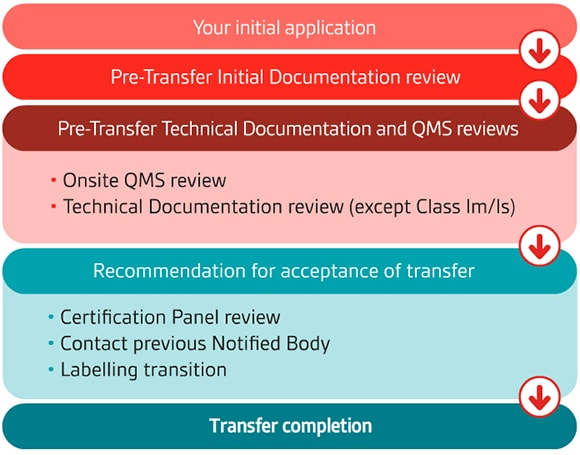




At BSI, we understand that having confidence in your Notified Body to deliver an efficient and robust CE marking and Quality Management System assessment process is important. Our approach focuses on open communication from the very beginning. Your application will be supported by a dedicated team of medical device experts.
We can offer a seamless service with comprehensive support and the absolute minimum level of disruption.