EN 60601 and Conformity Assessments
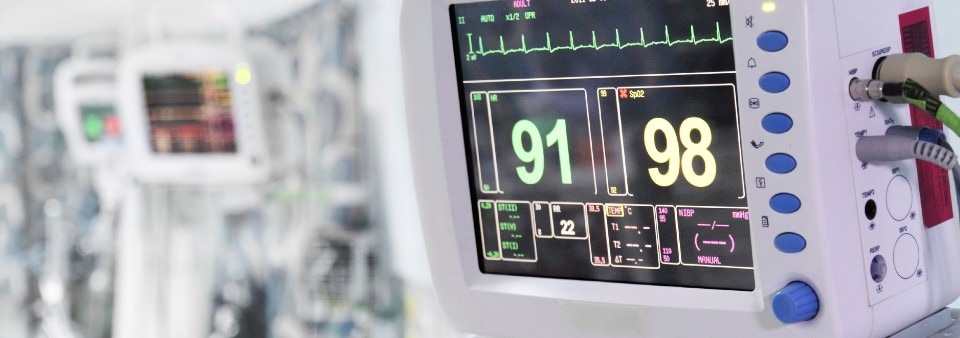
Manufacturers who wish to place medical electrical equipment onto the European or Great Britain market must obtain CE or UKCA Certification and apply the CE/UKCA mark on their device to demonstrate compliance with the appropriate legislation.
Whilst compliance with the state of art standards is not mandatory for CE/UKCA marking medical electrical equipment, this is the preferred method to demonstrate compliance with the applicable General Safety and Performance Requirements (GSPRs) of the Medical Devices Regulation (MDR) (EU) 2017/745 or with the Essential Requirements (ER) of UK Regulation. As few standards are currently harmonized to the regulations, for manufacturers of medical electrical equipment to be assessed against the Regulation, understanding and meeting the requirements of EN 60601 is crucial. EN 60601 represents a state-of-the-art standard and can therefore be applied to show conformity with the applicable MDR and UK Regulation requirements