What is an ophthalmic medical device?
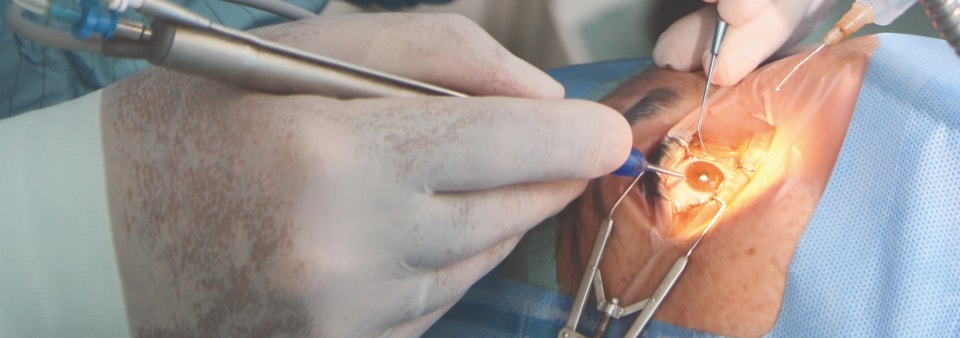
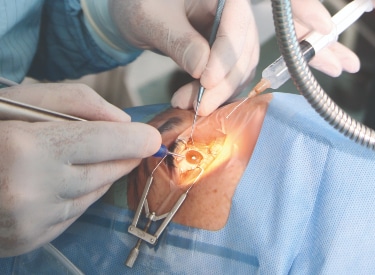
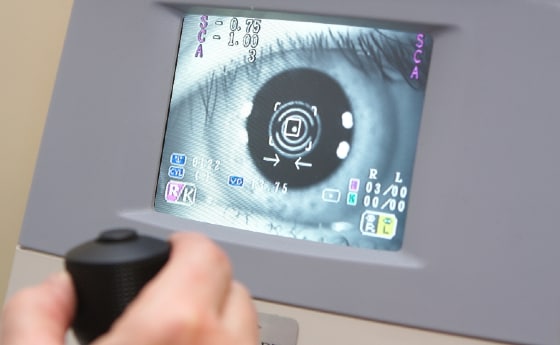
An ophthalmic medical device can be defined as a device that fulfils a medical purpose for use in optometry and ophthalmology. These devices range from non-invasive devices and instruments often used for diagnoses, to invasive devices, such as contact lenses (and their associated care products), and implantable devices, such as intraocular lenses and glaucoma stents. Surgical systems, including lasers and phacoemulsification machines, and surgical instruments are also examples of these devices.
We collaborate with our in-house clinicians and, where products require additional expertise, we consultant our other technical teams covering all areas from active and active implantable, to medicinal substances, devices utilizing animal tissue and sterile medical devices.
BSI is able to offer CE certification services under the EU MDR and UKCA services under the UK MDR 2002 for ophthalmic medical devices. Examples of products we cover include:
- Contact lenses (including cosmetic lenses)
- Contact lens care products
- Diagnostics
- Implants (including for the treatment of conditions of the cornea and glaucoma)
- Intraocular lenses
- Lasers
- Phacoemulsification systems
- Solutions (including artificial tears)
- Surgical instruments
For more clarity on ophthalmic and related medical devices, please refer to the MDR (EU) 2017/745 and the UK MDR 2002.